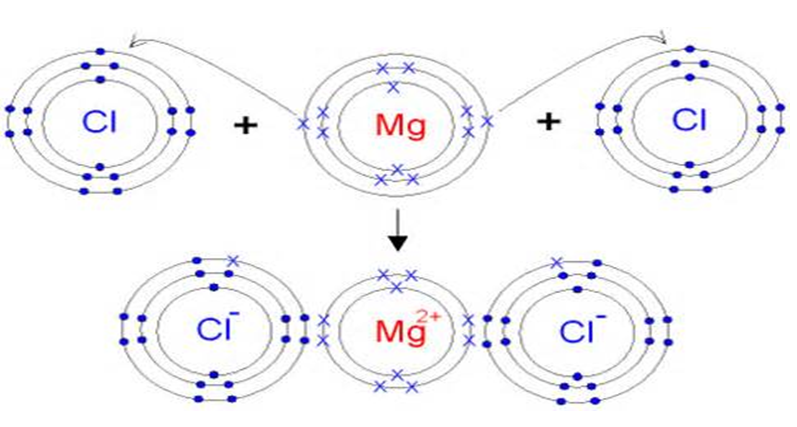
And so what I'm drawing right over here, these are ion-dipole forces. These water molecules so that you have the partial negative end is facing towards this The partial negative end ofĪ dipole would be attracted to a positively charged ion. So now that we know the differenceīetween ions and dipoles, how might they interact? Well you might guessĬoulomb forces are at play. To form hydrogen bonds just have a very strong dipole moment, because you have hydrogenīonded to an oxygen, a nitrogen, and a fluorine, But, as we've talkedĪbout, hydrogen bonds, which are an intermolecular force are just a special case of dipole forces. The partial positive end of another water molecule. And we describe this when we talked about hydrogen bonding where the partial negativeĮnd of one water molecule would be attracted to More time around the oxygen than around the hydrogen, you have a partial negative charge at this end of the molecule and you have partial positive charges at the other end of the molecules. Much more electronegative that it hogs the electrons, Is quite electronegative, covalently bonded to two hydrogens, and those are really polar covalent bonds because the oxygen's so Molecule that is a dipole or has a dipole moment atĪ molecular level is water. Where the charge is separated on different ends of the molecule, that you have a partially positive end and you have a partial negative end, that there is a molecular dipole moment. We're talking about dipoles, we're not talking about something that has necessarily a net charge, we're talking about something Now what's the differenceīetween that and a dipole? Well, generally speaking when Similarly, when sodium loses an electron, it now has a net positive charge. So for example, whenĬhlorine gains an electron and becomes the chloride ion, it's an ion because it now All right, now let'sįirst think about ions. And I encourage you to pause this video and try to refresh your own memory before we refresh our memories together. And before we think about how ions and dipoles might interact, let's just remind ourselves what the difference isīetween ions and dipoles.  
Let's talk a little bit about ion-dipole forces.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
